Thin Layer Chromatography Interview Questions and answers:
Thin-layer chromatography is a separation technique used in the pharmaceutical industry for identification tests or for related substance tests.
Q1: What is the Principle of Thin Layer Chromatography (TLC)?
A: Thin layer Chromatography (TLC) is a separation technique in which a stationary phase consisting of an appropriate material is spread in a uniform layer on the support of a plate (Glass, Metal, or Plastic).
Solutions of analyte deposited on the plate prior to development. The separation is based on adsorption, partition, Ion-exchange and on a combination of these mechanisms and is carried out by migration of solutes (Solution of the analyte) in a solvent or a suitable mixture of solvents (mobile phase) through the thin layer.
Q2: What is the definition of Adsorption?
A: Process in which a gas, liquid, or solid adheres to the surface of a solid or a liquid but does penetrate it.
Q3: What is the definition of Partition?
A: It is a vertical structure that divides or separates.
Q4: What is the definition of Ion-Exchange?
A: Process in which ions of one substance are replaced by similarly charged ions of another substance.
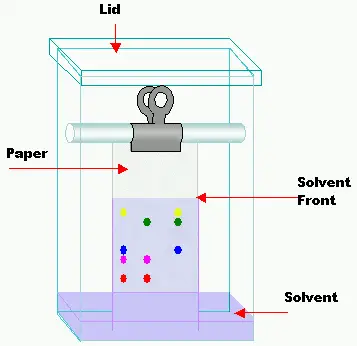
Q5: What is the vertical development of the chamber?
A: It is a tank with flat bottom or twin trough, of inert, transparent material, of a size suitable for the plate used and provided with a tightly fitting lid is required.
Q6: What is the horizontal development of the chamber? (As per EB/BP)
A: The tank is provided with a trough for a mobile phase and it additionally contains a device for directing the mobile phase to the stationary phase.
Q7: What is used for a stationary phase in TLC?
A: Silica gel and alumina are the most common stationary phases used in TLC but others are also available.
Q8: How to develop a TLC chamber?
A: Prepare the chamber by lining the walls with sheets or filter paper, pour the solvent into the tank, saturate the filter paper in the process, add sufficient mobile phase to form a layer of solvent 5-10mm deep, close the chamber and allow to saturate for 1 hour at room temperature.
Q9: How to apply sample on TLC plate?
A: Apply the solution on the plate at a suitable distance from the lower edge and from the side of the plate and on a line parallel to the lower edge.
Q10: How to do a spot–on TLC plate?
A: Draw a straight line across the plate with a pencil (do not use a pen) approximately 1 cm away from the bottom. Do not use excessive force while spotting. The spot should be applied in small time intervals (Around 1 min).
Q11: What should be the distance between two spots?
A: The distance between two spots should be at least 10mm.
Q12: What is the next procedure after sample application?
A: Once the sample applies on a plate in a circular manner with at least 10 mm distance, place the plate into the developed chamber which is filled by mobile phase, and wait till the mobile phase moved to 3/4 of the plate. spray the plate if required. Dry the plate in the oven or as specified in the method and visualize the plate in a visualizer or manual.
Q13: Which detector is used in TLC for visualizing?
A: UV detector used for visualizing.
Q14: What are the wavelengths used for TLC in pharma?
A: There are two wavelengths used for TLC; short-range 254 nm and long-range 365 nm.
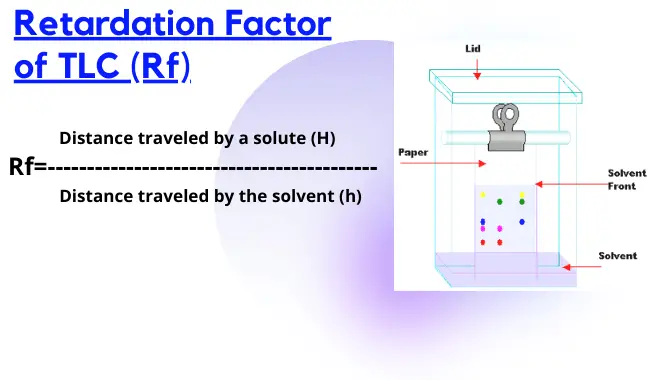
Q15: What is the Retardation factor (Rf) in TLC?
A: The retardation factor is a ratio of the distance from a point of application to the center of the spot and the distance traveled by the solvent front from the point of application.
OR
The distance traveled by a solute (Sample solution) to the distance traveled by solvent.
Q16: How to calculate the Rf value in TLC?
A: Rf= H/h
where, Rf= Retardation factor
H= solute distance
h= solvent front distance
Q17: What are the common problems in TLC?
A:
- Over large spots,
- Improper spotting,
- Streaking (Not in a single spot),
- The uneven advance of the solvent front
Thin Layer Chromatography Interview Questions will help you to clear your interview without fear.
Also Read:
- Karl Fischer Titration Interview Question and Answer
- Interview Question and answer on Polarimeter
- Dissolution Interview Question and Answer
- Gas Chromatography Interview questions and answers
- Requirement for conducting stability study in pharma
- Preparation and standardization of 1N NaOH as per USP
For interview preparation refer YT channel: Pharmabeejpro